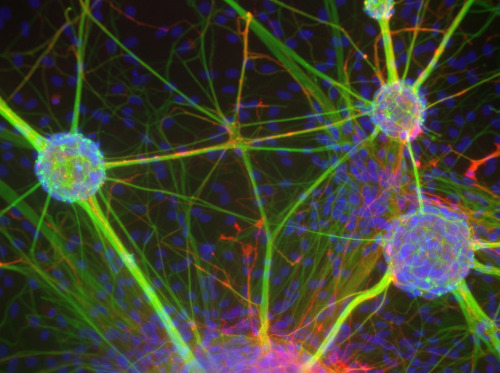
Моей радости нет предела - ещё одна новость об оксидативном повреждении ДНК, на http://www.eurekalert.org/pub_releases/2009-03/eu-drm032609.php
Public release date: 26-Mar-2009
Contact: Holly Korschun
hkorsch@emory.edu
404-727-3990
Emory University
Like doctors making house calls, some DNA repair enzymes can relocate to the part of the cell that needs their help, a collaborative team of scientists at Emory University School of Medicine has found.
The signal that prompts relocation is oxidative stress, an imbalance of cellular metabolism connected with several human diseases.
The study integrated the expertise of three Emory groups and resulted in a new level of understanding of the cell's response to genetic damage. The finding could lead to new targets for anti-cancer drugs that interfere with DNA repair, says Paul Doetsch, PhD, professor of biochemistry, radiation oncology, and hematology and oncology at Emory University School of Medicine.
The results were published in the February 1 issue of Molecular and Cellular Biology. The journal's editors chose an image of yeast cells with fluorescent DNA repair enzymes for the cover.
"DNA damage and oxidative stress are very closely related," Doetsch says. "For example, the way radiation inflicts most of its damage on DNA is through oxidative stress. The more we know about how cells respond to oxidative stress, the more chances there could be to influence those responses for diagnostic or therapeutic purposes."
The DNA inside cells is continually under assault by heat, radiation and oxygen. Cells have an extensive set of repair enzymes that comb through DNA, continually excising and re-copying damaged segments. To complicate matters, mitochondria (cells' miniature power plants) have their own DNA.
Working with Doetsch, Emory graduate students Lyra Griffiths and Dan Swartzlander, and biochemists Anita Corbett and Keith Wilkinson, genetically modified strains of yeast so that two different DNA repair enzymes would be fluorescent. They were able to follow the enzymes around the cell when yeast was exposed to hydrogen peroxide, causing oxidative stress, or to other chemicals causing DNA damage.
One DNA repair enzyme they studied, Ntg1, moves to the nucleus or the mitochondria depending on where DNA damage is concentrated, the authors found. In contrast, a related enzyme, Ntg2, stays in the nucleus under all conditions.
Cells appear to direct Ntg1's relocation by briefly attaching a small protein called SUMO to what needs to be moved around, the authors found. SUMO is found in fungi, plants and animals and is already being investigated by several research groups as a possible target for anti-cancer drugs.
The Robert W. Woodruff Health Sciences Center of Emory University is an academic health science and service center focused on missions of teaching, research, health care and public service. Its components include schools of medicine, nursing, and public health; the Yerkes National Primate Research Center; the Emory Winship Cancer Institute; and Emory Healthcare, the largest, most comprehensive health system in Georgia. The Woodruff Health Sciences Center has a $2.3 billion budget, 17,000 employees, 2,300 full-time and 1,900 affiliated faculty, 4,300 students and trainees, and a $4.9 billion economic impact on metro Atlanta.